Mission Health Communities Enhances Resident Care Through eBrief™ Early Adopter Program
UF Innovate | Accelerate resident client etectRx™ announces its partnership with Mission Health Communities, marking a significant addition to the growing roster of eBrief™ partners.
Podcast: What’s Next Longevity Deal Talk – etectRx & Trilogy Health
UF Innovate | The Hub resident company etectRx, Inc., is partnering with Trilogy Health to pilot its latest technology, designed to assist caregivers and older adults in managing urinary incontinence. Eric Buffkin, CEO of etectRx, join Mary Furlong & Associates’ podcast to discuss how this innovative solution can prevent falls during the night and support staffing.
Trilogy Health Services Joins eBrief™ Early Adopter Program
UF Innovate | Accelerate resident client etectRx™ announced an eBrief Early Adopter Program (EAP) partnership with Trilogy Health Services, a senior care communities thought leader and provider.
Tech Tuesday – etectRx
Tech Tuesday with Eric Buffkin, CEO of etectRx, as he outlines the company’s newest partnership with the Village in Gainesville to improve incontinence monitoring, elevate care for seniors, and alleviate pressure on staff.
etectRx and the Village at Gainesville Announce Intelligent Incontinence Care Collaboration
UF Innovate client etectRX recently announced a first-of-its-kind pilot project with The Village at Gainesville, a vibrant retirement community. The project will center around the company’s “eBrief” technology, a patented intelligent incontinence monitoring system.
ID-Cap™ System Powers Historic HIV Digital Pill Study
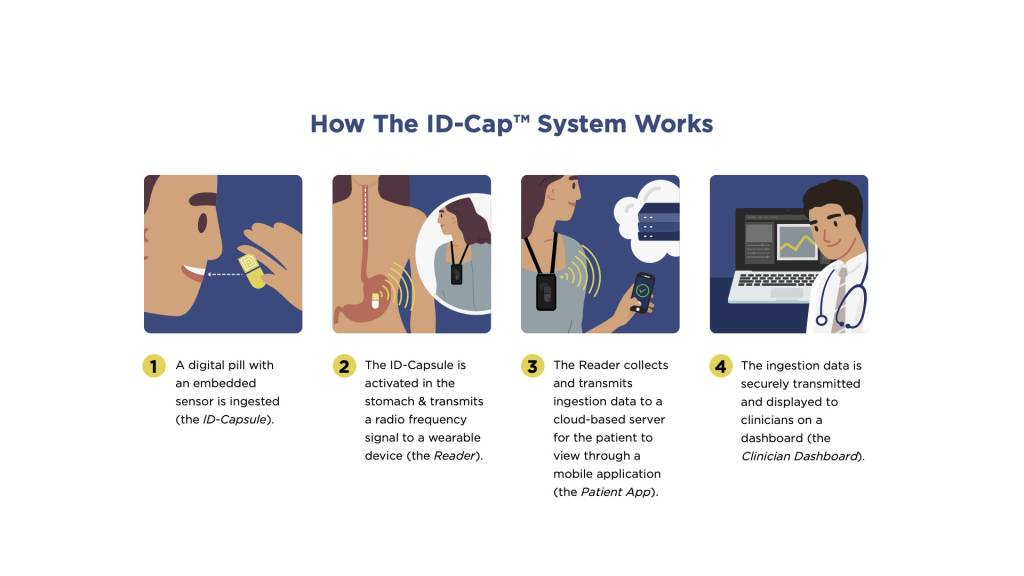
etectRx announces record-breaking use of the ID-Cap™ System, utilized in University of Colorado’s HIV treatment study.
Why Aren’t Digital Pills Taking Off?
In 2017, the US Food and Drug Administration (FDA) broke ground by approving the first drug with an embedded biosensor to track its use. The enthusiasm for digital approaches involving sensors, apps, and wearables that could transmit information across systems, commonly grouped under the term Internet of Things, also spread. Yet, despite the landmark FDA approval, digital pills have not exploded in pharma. Privacy and logistical concerns, especially while studying such applications for vulnerable populations, have lingered.
Medication Safety 2.0: Advances in Adherence Technology
A recently published review on emerging smart technology innovations in the health care sector suggests various advanced technologies and methods may now significantly improve medication adherence.1 Two significant advances have been the ability to measure medication adherence and safety issues with medication event monitoring systems (MEMS) and smart blister packs containing radio frequency identification technology.
etectRx Announces Eric Buffkin as New CEO
UF Innovate | The Hub resident etectRx, Inc., a privately-held digital health company, announced the appointment of Eric Buffkin as its next president and CEO.
etectRx Announces Launch of MyTPill Study To Address Medication Adherence Among HIV+ Prescription Opioid Users
UF Innovate | The Hub resident company etectRx, Inc., a privately-held digital health company, announced the launch of a study to monitor antiretroviral therapy adherence among prescription opioid users living with HIV.
