Inside LPRC IMPACT 2026

A cool opening-night reception at UF Innovate | The Hub and the LPRC INNOVATE labs marked the start of a learning-packed few days at the 2026 LPRC IMPACT Conference, March 17-19.
Oragenics Receives HREC Approval for Phase IIa Clinical Trial of ONP-002 as a Treatment for Concussion and Mild Traumatic Brain Injury in Australia (Globe Newswire)

UF Innovate | Accelerate graduate Oragenics has received final ethics approval in Australia to begin its Phase IIa clinical trial of ONP-002, an intranasal therapy being developed to treat concussion and mild traumatic brain injury.
Verizon Is Investing in a New Generation of Life-Saving Disaster Resilience Technology (Verizon)

UF Innovate | Accelerate resident client FNN has been selected as one of four innovators in the 2025 Verizon Disaster Resilience Prize, a $1 million program supporting technologies that help communities better prepare for and respond to natural disasters.
Oragenics Targets Projected $9 Billion Market by Advancing First and Only Clinical-Stage Concussion and Mild Traumatic Brain Injury Therapy (Globe Newswire)

UF Innovate | Accelerate graduate Oragenics, Inc. outlined its 2026 milestones as it advances ONP-002, a novel intranasal neurosteroid in clinical development for concussion and mild traumatic brain injury.
Solid Biosciences Announces Positive Feedback From Type C Meeting With FDA for SGT-003 Gene Therapy for Duchenne Muscular Dystrophy (Globe Newswire)

Solid Biosciences Inc., which acquired UF startup AavantiBio, announced a positive regulatory update from its recent Type C meeting with the U.S. FDA that supports the continued advancement of SGT-003 as a potential treatment for Duchenne muscular dystrophy.
Oragenics Partners With DUCK FLATS Pharma To Support FDA IND Readiness and Clinical Trial Design for Concussion Program (Globe Newswire)

UF Innovate | Accelerate graduate Oragenics announced it has engaged DUCK FLATS Pharma as its U.S. Investigational New Drug (IND) readiness and regulatory execution partner to support FDA-facing preparation and clinical trial design as the Company advances its novel intranasal concussion therapy toward U.S.-based development.
Amend Surgical Announces ISO 13485 Certification, Advancing Toward Launch of Amend Tissue Tape™ (PR Newswire)
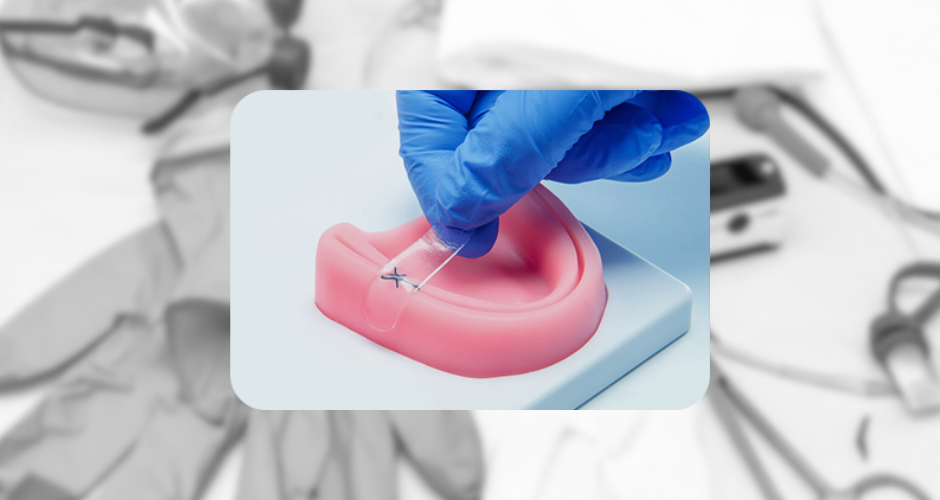
UF Innovate | Accelerate client Amend Surgical announced that it has achieved ISO 13485:2016 certification, the global standard for medical device quality management systems.
Mark Cuban Cost Plus Drugs and Alchem Laboratories Announce Alliance To Advance Essential Medicines (EIN Newswire)

UF Innovate client Alchem Laboratories announced a partnership with Mark Cuban Cost Plus Drugs to support the development and delivery of essential medicines.
Solid Biosciences Announces Duchenne Muscular Dystrophy Added to National Recommended Uniform Screening Panel by the U.S. Department of Health and Human Services (Bio Space)

Solid Biosciences Inc., which acquired UF startup AavantiBio, shared that the U.S. Department of Health and Human Services (HHS) officially added Duchenne muscular dystrophy (Duchenne) to the Recommended Uniform Screening Panel (RUSP), the list of conditions recommended for universal newborn screening across the United States.
TuHURA Biosciences Announces Its Release of Kintara’s Contingent Value Right (CVR) As Kintara’s REM-001 Meets Primary Safety Endpoint Achieving Contractual Milestone (PR Newswire))

TuHURA Biosciences, formed by CohBar and Morphogenesis, a UF startup, announced that Kintara’s REM-001 clinical trial of ten metastatic cutaneous breast cancer patients met its primary endpoint, demonstrating safety with signs of clinical efficacy following eight weeks of follow-up for such patients.
